And then Carbon, in the center, needs 8, and it has 8. Difluoromethane, also called difluoromethylene, HFC-32 Methylene Fluoride or R-32, is an organic compound of the dihalogenoalkane variety. Fluorine needs 8, this Fluorine has 8 and this Fluorine has 8. 
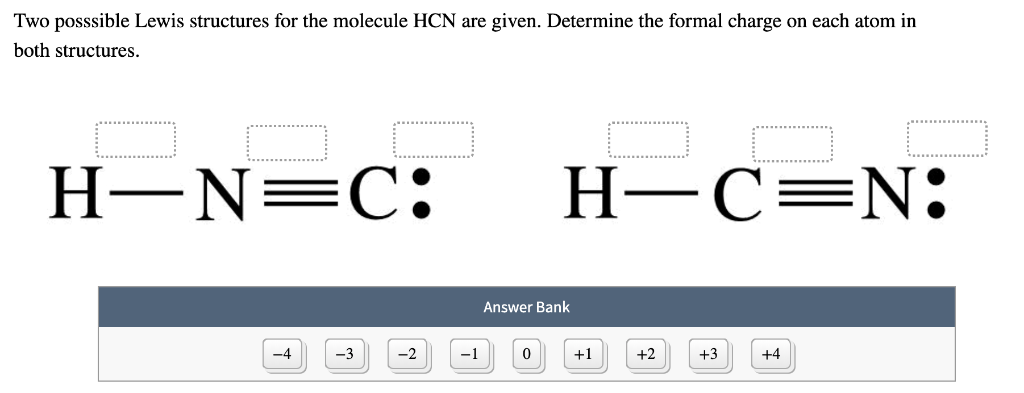
Hydrogen only needs two valence electrons, so it's OK. We'll start by putting two valence electrons between each of the atoms to form a chemical bond, and that's used up 2, 4, 6, 8. And the Fluorines are very electronegative so they go on the outside, as well. Study with Quizlet and memorize flashcards containing terms like Which of the following takes up the most space Answers: Double bond Lone pair of electons Triple bond Single bond, How many sigma bonds are in ethene, C2H4 Answers: 5 4 6 1, How many pi bonds are in carbon monoxide, CO Answers: 3 1 2 0 and more. We can describe the molecular geometry of HCN by knowing first the central atom for the compound. Molecular Shape, Hybridization & Polarity CH2F2 Lewis dot structure, Shape. We'll put the Carbon at the center, and then Hydrogens always go on the outside. Molecular Geometry: The given compound is hydrogen cyanide (HCN). BOND ANGLE(S) MOLECULE ELECTRON GEOMETRY MOLECULAR GEOMETRY CS HCN CHO SO2 CHA. Fluorine, 7 valence electrons, we have 2 of those as well, for a total of 20 valence electrons. Hydrogen, group 1, but we have 2 Hydrogens. As a result, the CO 2 molecule has no net dipole moment even though it has a substantial separation of charge. Because the two CO bond dipoles in CO 2 are equal in magnitude and oriented at 180 to each other, they cancel. The hybridization number of CF4 is Sp and the steric number is 4. The overall formal charge in CF4 is zero. CF4 molecular geometry is tetrahedral and its electron geometry is also tetrahedral. On the periodic table, Carbon is in group 4, it has 4 valence electrons. Each CO bond in CO 2 is polar, yet experiments show that the CO 2 molecule has no dipole moment. A total of 8 bonded electrons are present in the CF4 lewis dot structure. Let's do the Lewis structure for CH2F2: difluoromethane. This gives us (Adapted from Quora) Step 2. We have '7 + 2×7 -1 20' electrons to fit around the atoms so that every atom gets an octet. HCN and CH2O4 obitan lai ha sp3 xen ph vi obitan 1s ca nguyn t. Draw the Lewis structure We put the least electronegative atom in the centre. CH2F2 Molecule : Determine the form of CH2F2 molecular geometry using VSEPR theory. With fewer 90° LP–BP repulsions, we can predict that the structure with the lone pair of electrons in the equatorial position is more stable than the one with the lone pair in the axial position.Transcript: Hi, this is Dr. The molecular shape is 'bent', with a theoretical bond angle of 109.5°. If we place it in the axial position, we have two 90° LP–BP repulsions at 90°. In the CH 2 F 2 lewis structure, a total of 6 lone pairs and 4 bond pairs are present. The molecular geometry or shape of CH 2 F 2 is tetrahedral. However, because the axial and equatorial positions are not chemically equivalent, where do we place the lone pair? If we place the lone pair in the equatorial position, we have three LP–BP repulsions at 90°. The approx bond angle in CH 2 F 2 is based on the type of bond, F-C-H 109º, H-C-H 113º, F-C-F 108.5º. We designate SF 4 as AX 4E it has a total of five electron pairs. structure was found for X HCF3 and NH3, while HCNHCNCO 2 is T-shaped and HCNHCNH 2O is an asymmetric top.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
